It took me three months to convince Ms. Q for the “Yuk” treatment, as she understood fecal microbiota transplantation. She had suffered multiple relapses of C. difficult infection (CDI) requiring hospitalization. She had developed profound protein energy malnutrition, was depressed and had lost the ‘will’ to go on like this. Desperate, she agreed. “Hell, I’ll do anything to get rid of this,’ ”
FMT has proved highly effective at eradicating C. difficile infection and restoring a healthy gut microbiota. Cure rates are close to 95% for relapsing CDI, and severe infections. It is proven to be safe and easily accomplished by one of the following ways – (top vs bottom) nasogastric tube , retention enemas or via colonoscopy. As expected, Ms Q liked the bottom approach via colonoscopy.
Since, this was my first FMT a protocol was developed as detailed below. Prior to FMT, Ms. Q was started on TPN to bolster her nutrition and immunity.
1. Donor selection : like any other transplant, FMT needs a healthy donor. The donor is screened for infectious diseases like hepatitis C, B and HIV. In Ms Q’s case, her brother was the donor. Though I do not know of any case where feces have been implicated as the agent to transmit the above viruses.
2. Collection of the specimen : the specimen should be fresh with no additives added. Her brother came to the hospital the day of the procedure, the specimen was collected in a stool hat, sealed and transported to endoscopy suite in a bio-hazard bag.
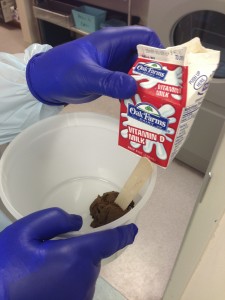
3. Specimen processing: 25 gm of feces was emulsified with milk and water in a closed lid container. The consistency was achieved so that the specimen could be dispersed through the channel of the colonoscope.
4. Delivery : the processed specimen was delivered to the entire colon with 50 cc syringes. A total of 200 cc emulsified feces were dispersed in the colon.
The whole process went smoothly and is a tribute to the nursing staff of Mainland Medical Center. The staff was attentive to the details, responsive and executed perfectly to enable me perform the transplant.
As with any pioneering treatment, there are hurdles that need to be cleared and FMT has gone through the same journey.
Early this summer in 2013, the FDA firmly declared poop a “biologic” and warned doctors they must acquire an “Investigational New Drug” research permit to administer it. However, doctors pounded the FDA. They complained that having to go through a cumbersome research process threatened to add costs and limit access for seriously ill patients. Just two months later, the agency relented, saying doctors didn’t need a permit to administer a fecal transplant to patients with recurrent C. diff.
I believe there remains quite a few hurdles that we need to cross to make it a successful and the treatment of choice for recurrent/relapsing CDI and seriously ill patients. I reiterate it is gross and ‘Yuk” treatment, but has proven to be the most effective way of eradicating the infection.
The FMT has not gained traction because it lacks any corporate interests, and on the contrary would be detrimental to the businesses of pharmaceuticals promoting the anti C Difficile antibiotics.
I personally would like to start a project of educating my fellow colleagues about the prospects of FMT and choosing it as the treatment of choice for the recurrent CDI and seriously ill patients.
While leaving hospital fully treated, I told Ms. Q that FMT is an inspirational treatment, just like the beautiful flower Lotus which arises from the dirty water.
If you wish to see me, please schedule a visit at one of the locations : 281-557-2527
- Clear Lake Office (Wesbter)
1015 Medical Center Blvd
Suite 1300, Webster, TX 77598
Phone: (281) 557-2527
- Satellite Office 2
7111 Medical Center Drive
2nd Floor
Texas City, TX 7759
If you have questions please do not hesitate to call my office 281-557-2527, and I would be more than happy to call you back and talk to you for free. I believe this is the least I can do to better the lives of people who are trying to find the right answers.
 Follow
Follow
No comment